With continuous innovation, Hana Pharm advances R&D excellence
and technological value, contributing to human health
through the development of superior pharmaceuticals.

Hana Pharm’s Research Division is strengthening its R&D capabilities by securing outstanding research talent and establishing advanced research infrastructure in order to evolve into an R&D-driven pharmaceutical company.
Through the activation of industry–academia–research collaboration, the division conducts a wide range of research and development projects. In particular, Hana Pharm possesses outstanding
technological expertise in the fields of sedatives/anesthetics and pain management, and demonstrates strong research capabilities across active pharmaceutical ingredient (API) synthesis,
technology-driven improved new drugs, differentiated generic medicines, and novel drug development, thereby playing a leading role in the pharmaceutical industry.
Furthermore, to create synergy based on its core strengths, Hana Pharm’s Research Division is developing next-generation innovative medicines, including the general anesthetic Byfavo Injection and treatments
for gastroesophageal reflux disease (GERD).
Looking ahead, Hana Pharm will continue to build a world-class research environment through focused investment in R&D, strengthening its competitiveness to emerge as a global pharmaceutical company.

Since its establishment, Hana Pharm has focused on securing advanced facilities and technologies for the stable production of high-quality active pharmaceutical ingredients (APIs). Through these efforts, the company has obtained manufacturing approvals for more than 30 APIs, as well as multiple related patents.
Hana Pharm’s Research Division develops innovative and efficient synthesis technologies, with the goal of building high-quality, high value-added API manufacturing capabilities that ensure strong market competitiveness.
Leveraging these capabilities, Hana Pharm established a scalable manufacturing process for HN01001 at the research stage, enabling stable, high-quality production. The company successfully supplied kilogram-scale API for Phase I clinical trials and is now advancing toward commercial-scale manufacturing to support mass production and market competitiveness of innovative new drugs.
In addition, Hana Pharm developed a new synthesis process for Remimazolam besylate, the API of the novel general anesthetic Byfavo Injection, for domestic regulatory approval. The company plans to expand approvals to global markets, strengthening its position as a competitive global API supplier and driving sustainable international growth.

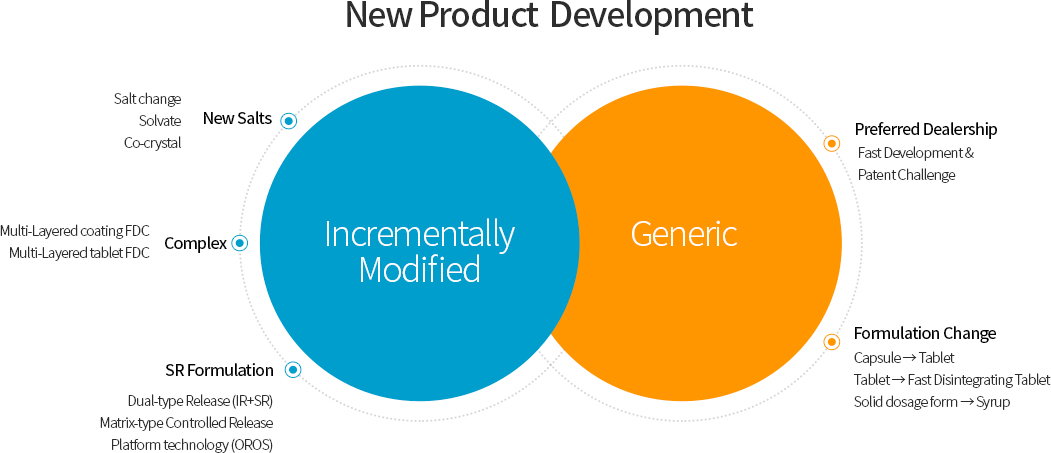
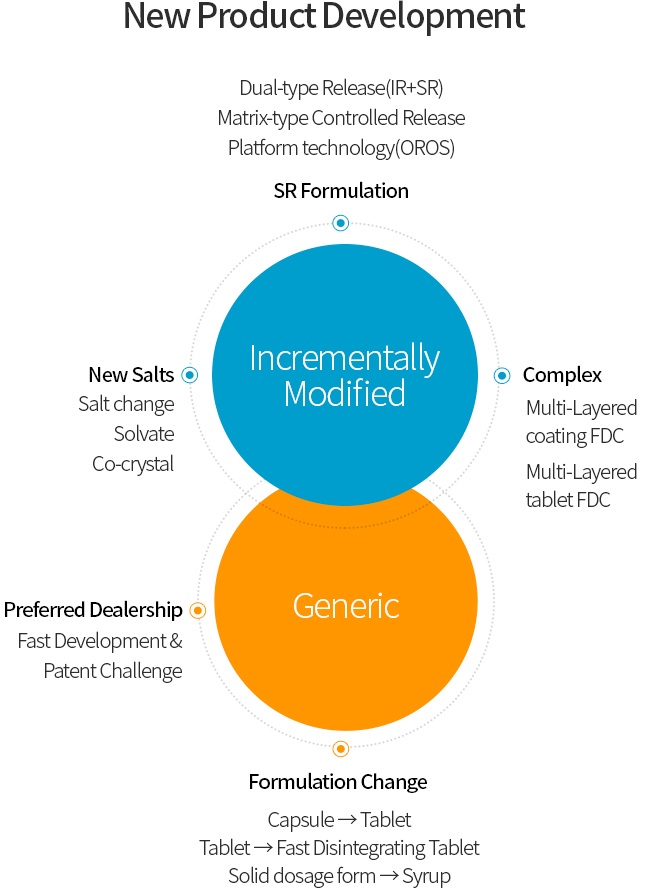
Hana Pharm’s Research Division is dedicated to the research and development of Incrementally Modified Drug(IMD) and Value-added Generic, with a strong focus on Drug Delivery System (DDS) technologies. These research achievements directly contribute to improving public health and serve as a key driving force behind the company’s sustainable revenue growth.
Hana Pharm possesses a wide range of advanced formulation technologies, including solubilization of poorly soluble drugs, controlled-release systems, and Fixed-Dose Combination(FDC) drug manufacturing. Leveraging these capabilities, the company is developing innovative pharmaceutical products that reduce adverse effects of existing drugs while enhancing patient convenience. As a result, from 2022 to 2025, Hana Pharm successfully obtained marketing approvals for many products and launched them into the market. Currently, Hana Pharm is actively conducting research on approximately 30 projects, including New Drug, Incrementally Modified Drug(IMD) , Value-added Generic.

Innovative drug development is actively pursued at Hana Pharm’s Pangyo R&D Center, supported by independent research capabilities and a systematic R&D process. Byfavo Injection, a next-generation sedative and general anesthetic, was in-licensed from PAION AG (Germany) in 2013. Hana Pharm subsequently conducted Phase III clinical trials for general anesthesia in the Republic of Korea and obtained new drug approval from the Korean Ministry of Food and Drug Safety (MFDS) in January 2021. HN01001, Hana Pharm’s proprietary drug candidate, is currently being developed for the treatment and maintenance therapy of gastroesophageal reflux disease (GERD). In addition, HN01001 is expected to provide effective therapeutic options for a broad range of gastrointestinal disorders, including Helicobacter pylori (H. pylori) eradication, Barrett’s esophagus, and peptic ulcer.